What Is A Property Of Most Molecular Compounds
Covalent compounds are the ones having strong intra-molecular bonds. The addition of HBr to either cis- or trans-2-butene is an example of racemic product formation the chiral center is colored red in the following equation.

What Is The Most Reactive Nonmetal And Why Name Symbols Inert Gas Noble Gas
NH 3 - ammonia.
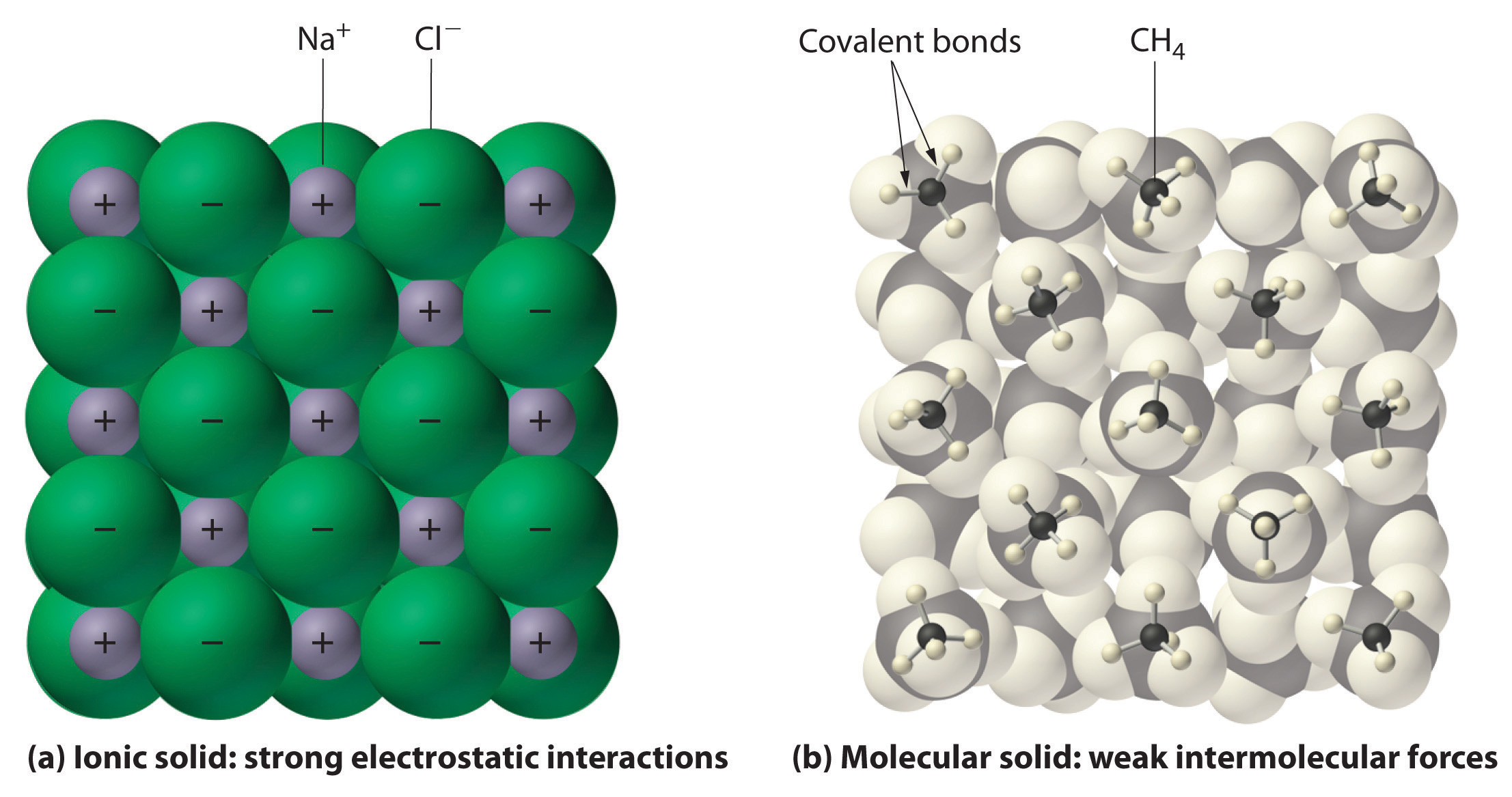
What is a property of most molecular compounds. The general MF query syntax consists of a series of valid atomic symbols please consult your periodical chart each optionally followed by either a number or a range. Pnictide compounds tend to be exotic. Binary compounds of the group can be referred to collectively as pnictides.
This is because the atoms within the covalent molecules are very tightly held together. Molecular Formula Input Molecular Formula MF -- Specification of the count of each element in a compound. CH 3 CO 2 H - acetic acid.
Weak electrolytes include weak acids weak bases and a variety of other compounds. Most compounds that contain nitrogen are weak electrolytes. H 2 O - water weakly dissociates in itself.
Each molecule is indeed quite separate and the force of attraction between the individual molecules in a covalent compound tends to be weak. When chiral compounds are created from achiral compounds the products are racemic unless a single enantiomer of a chiral co-reactant or catalyst is involved in the reaction. For example ethyl alcohol has the MF C 2 H 6 O.
HF - hydrofluoric acid. Miscibility m ɪ s ɪ ˈ b ɪ l ɪ t i is the property of two substances to mix in all proportions that is to fully dissolve in each other at any concentration forming a homogeneous mixture a solutionThe term is most often applied to liquids but also applies to solids and gasesFor example water and ethanol are miscible because they mix in all proportions. The most important elements of this group are nitrogen N which in its diatomic form is the principal component of air and phosphorus P which like nitrogen is essential to all known forms of life.

Phsc 1001 Matter Chart Teaching Chemistry Chemistry Basics Printable Worksheets
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry

Molecular Substance Definition Properties Video Lesson Transcript Study Com
What Defines A Molecular Compound Quora

6 2 Comparing Ionic And Molecular Substances Chemistry Libretexts

Clo2 Lewis Structure Chlorine Dioxide In 2021 Lewis Chlorine Molecules
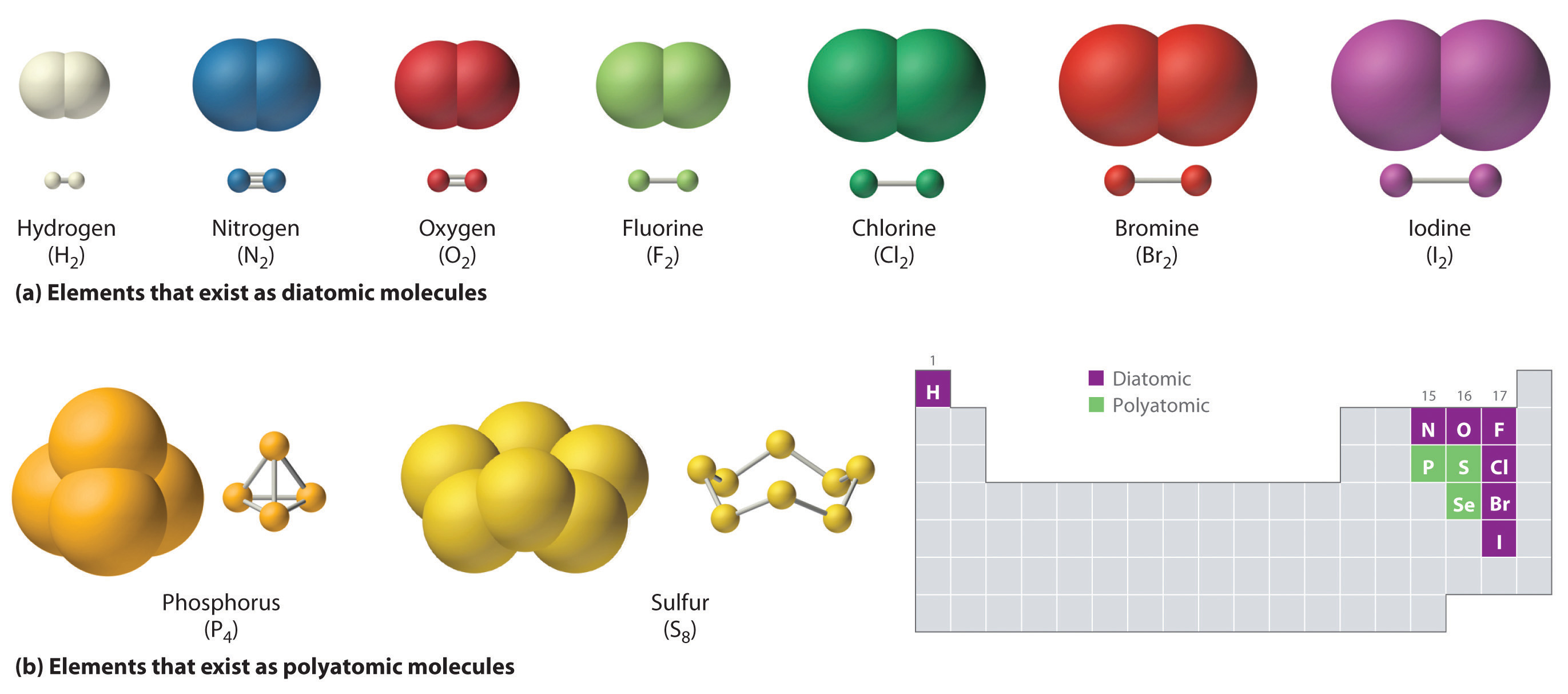
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry

Chemistry Physical Changes Physical And Chemical Properties Chemical Changes Chemical Reactions

What Are Hybrid Orbitals Master Organic Chemistry Organic Chemistry Chemistry Molecular Geometry

Is Hbr Polar Or Nonpolar Hydrogen Bromide In 2021 Molecules Hydrogen Atom Polar

Difference Between Empirical And Molecular Formula Infographic Chemistry Basics Chemistry Study Guide Chemistry Education

Ionic And Molecular Compounds Chemistry Escape Room Activity Chemistry Activities How To Memorize Things Molecular

Ionic Bonds And Ionic Compounds Ck 12 Foundation Chemistry Projects Chemistry Classroom Teaching Chemistry

Selina Concise Chemistry Class 8 Icse Solutions Chapter 3 Elements Compounds And Mixtures Ncert Books Ncertbooks Chemistry Class Chemistry Matter Science

Difference Between Organic And Inorganic Compounds Definition Structure Properties Chemistry Lessons Chemistry Education Study Chemistry

Ionic Bond Vs Covalent Bond Covalent Bonding Covalent Bonding Worksheet Ionic Bonding

Chemistry Lab Molecular Models Of Covalent Compounds Polar Or Nonpolar Chemistry Labs Chemistry Science Chemistry

Do Soy Burgers Processed Vitamins Contain Hexane Medicinal Chemistry Chemical Science Chemistry Labs
