What Are The Three Examples Of Covalent Bonds
Hybridization is a concept used in organic chemistry to explain the chemical bonding in cases where the valence bond theory does not provide satisfactory clarification. In a covalent bond the atoms are bound by shared electrons.
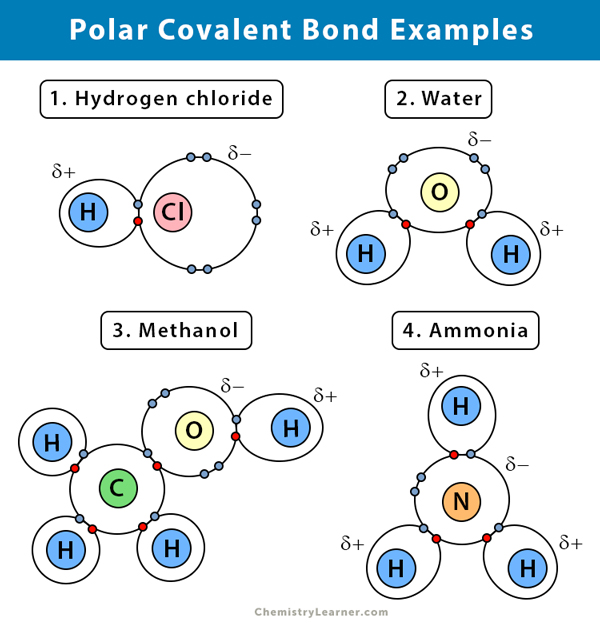
Polar Covalent Bond Definition And Examples
Usually an electron is more attracted to one.

What are the three examples of covalent bonds. But there are weak Van der Waal forces between these planar structures. Examples of compounds include table salt or sodium chloride NaCl an ionic compound sucrose a molecule nitrogen gas N 2 a covalent molecule a sample of copper intermetallic and water H 2 O a covalent molecule. Network CovalentThese bonds are created by nonmetal atoms just like molecular covalent bonding but they are made of solids.
In the formation of a nitrogen molecule each nitrogen atoms having five valence electrons provides three electrons to form three electron pairs for sharing. Electronegativity is probably the most important concept to understand in organic chemistry were going to use the definition that Linus Pauling gives in his book the nature of the chemical bond so Linus Pauling says that electronegativity refers to the power of an atom in a molecule to attract electrons to itself so if I look at a molecule Im going to compare two atoms in that molecule Im. Three dimensional configurations are best viewed with the aid of models.
Instead of the valence electrons being shared between the atoms in network covalent bonding the electrons are shared amongst the entire atoms. The three dimensional shape or configuration of a molecule is an important characteristic. Polar bonds are the dividing line between pure covalent bonding and pure ionic bondingPure covalent bonds nonpolar covalent bonds share electron pairs equally between atoms.
The most common examples of covalent network structures are graphite diamond quartz fullerene etc. These types of bonds do not conduct electricity nor do they dissolve in water. Technically nonpolar bonding only occurs when the atoms are identical to each other eg H 2 gas but chemists consider any bond between atoms with a difference in electronegativity less than 04 to be a.
In a true covalent bond the electronegativity values are the same eg H 2 O 3 although in practice the electronegativity values just need to be closeIf the electron is shared equally between the atoms forming a covalent bond then the bond is said to be nonpolar. In graphite one carbon atom is always bonded to three other carbon atoms via covalent bonds. Triple covalent bonds are represented by three dashes and are the least stable types of covalent bonds.
This gives graphite a complex. Therefore graphite has a planar structure. This theory is especially useful to explain the covalent bonds in organic molecules.
A chemical bond is a lasting attraction between atoms ions or molecules that enables the formation of chemical compoundsThe bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bondsThe strength of chemical bonds varies considerably. This shape is dependent on the preferred spatial orientation of covalent bonds to atoms having two or more bonding partners. There are strong bonds or primary bonds such as.
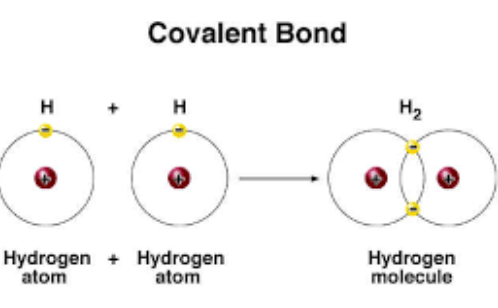
What Are The Types Of Covalent Bonds Science Online
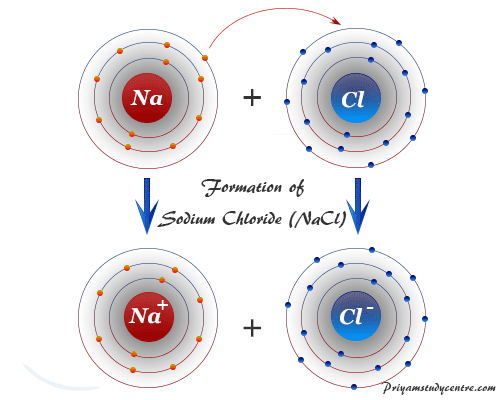
Covalent Bond Types Definition Properties Examples

Covalent Bond Images Stock Photos Vectors Shutterstock
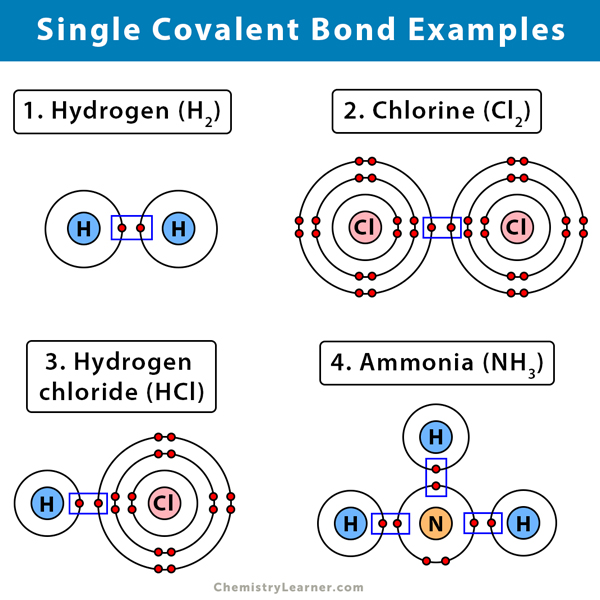
Single Covalent Bond Definition And Examples
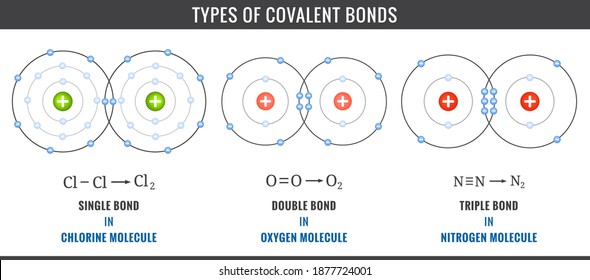
Covalent Bond Images Stock Photos Vectors Shutterstock
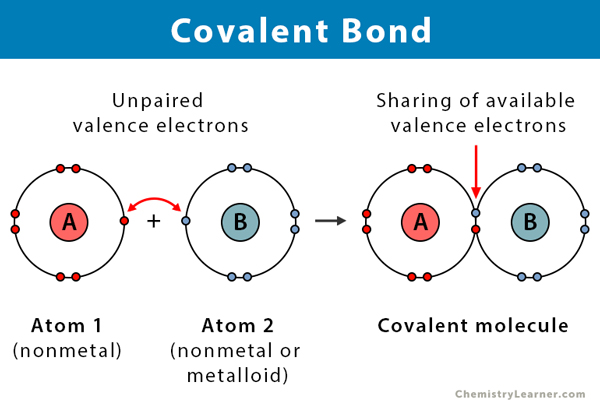
Covalent Bond Definition Types And Examples
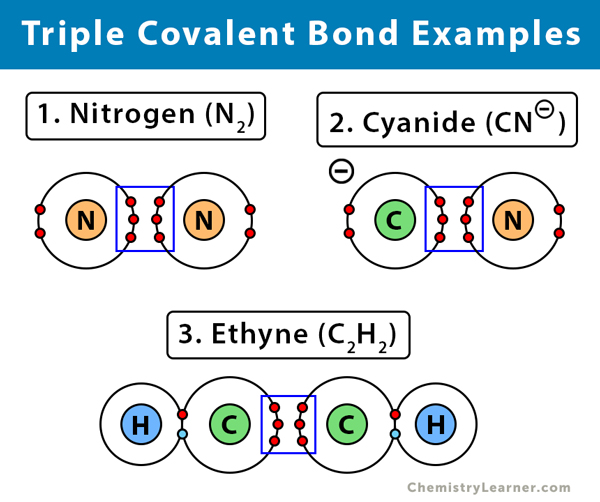
Triple Covalent Bond Definition And Examples
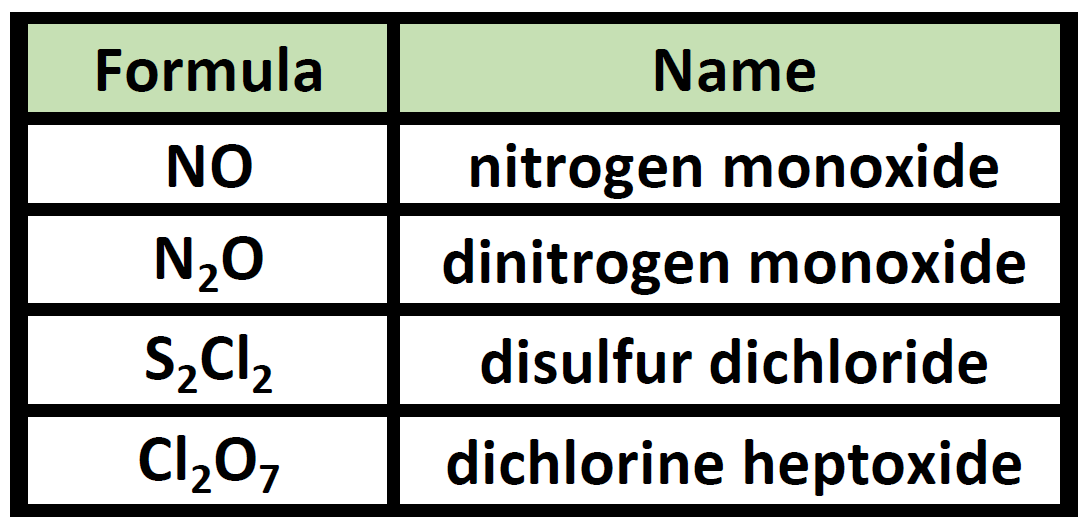
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry
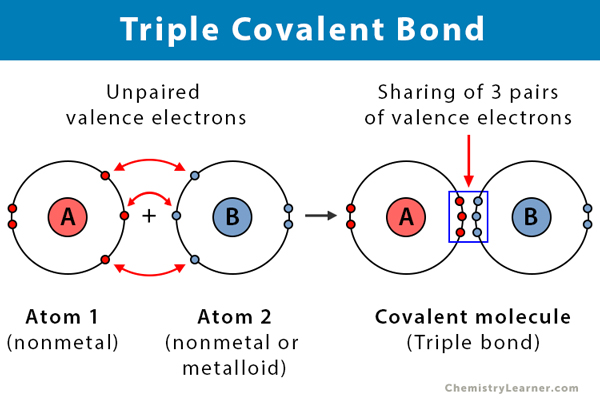
Triple Covalent Bond Definition And Examples
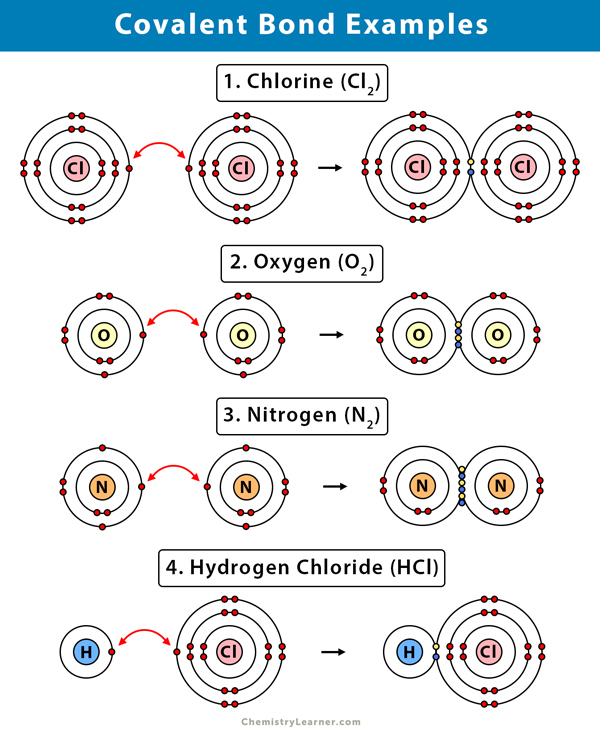
Covalent Bond Definition Types And Examples
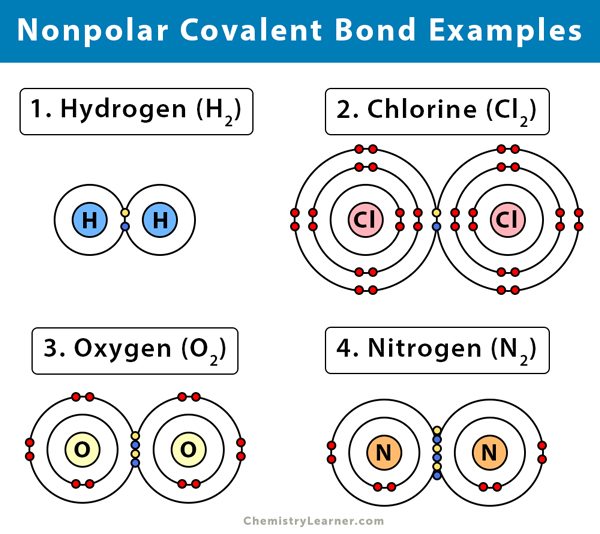
Nonpolar Covalent Bond Definition And Examples
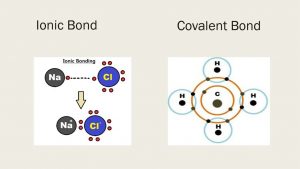
Chemical Bonds Ionic Bonds Properties Types Of Covalent Bonds Science Online

Covalent Bonds Biology For Majors I
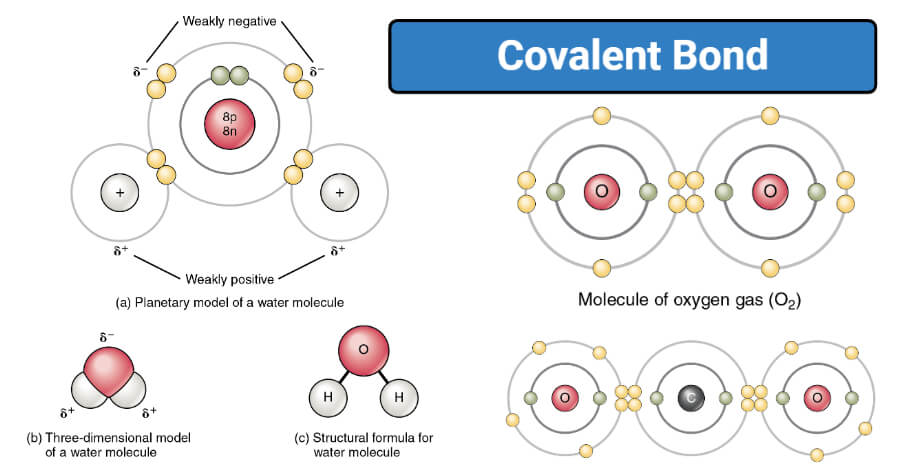
Covalent Bond Definition Properties Types Formation Examples
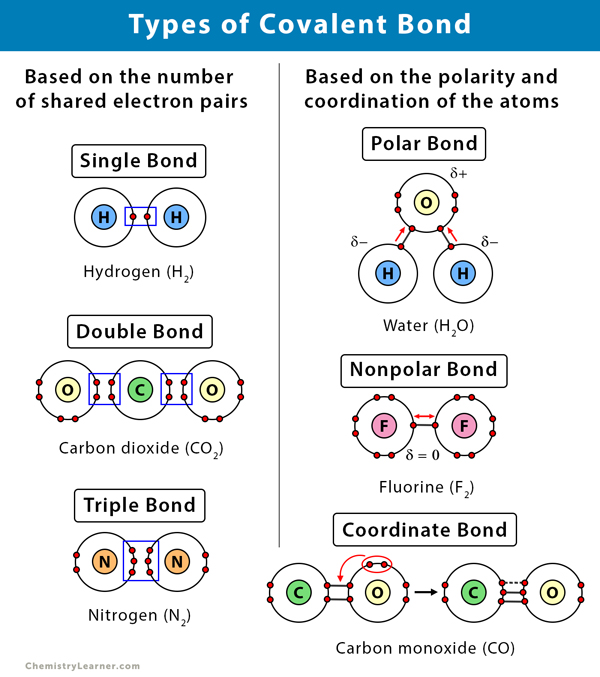
Covalent Bond Definition Types And Examples
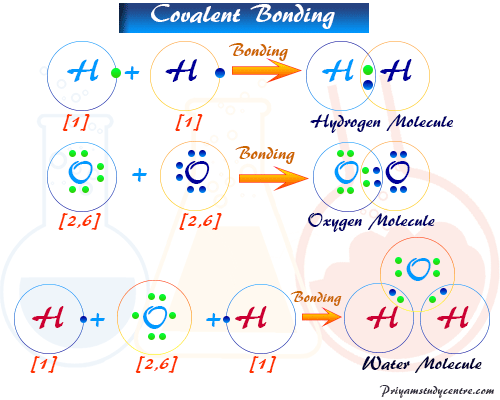
Covalent Bond Types Definition Properties Examples
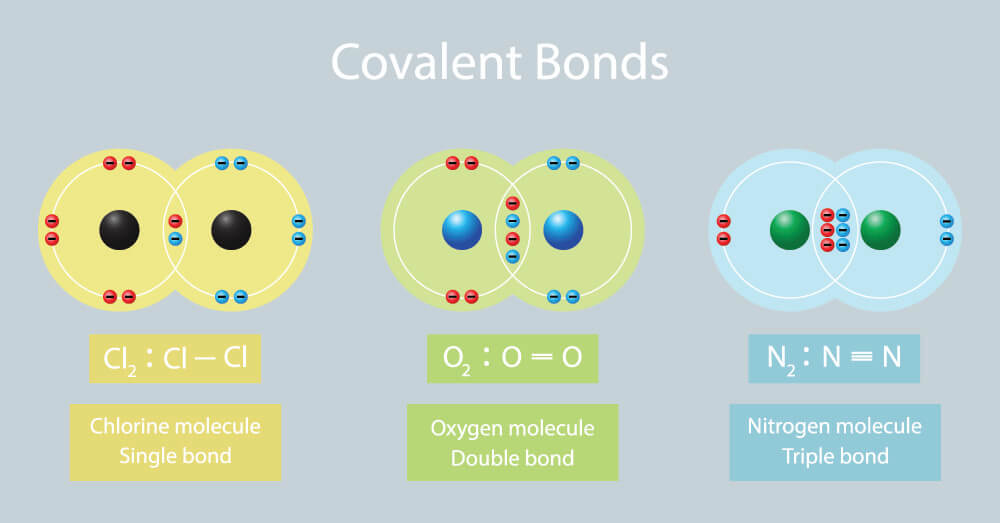
Covalent Bond Biology Dictionary

Covalent Bond Definition Properties Examples Facts Britannica
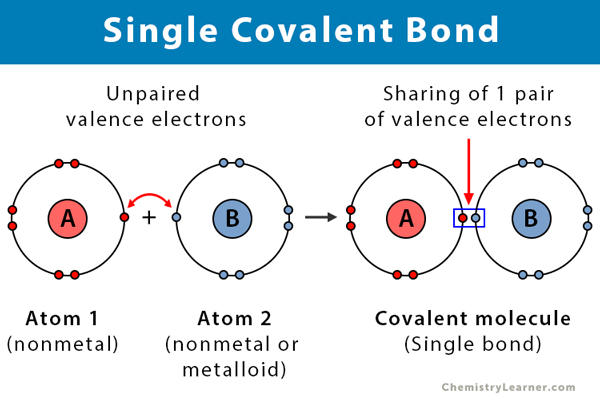
Single Covalent Bond Definition And Examples