What 2 Elements Make Up A Covalent Compound
The water molecule is made up of oxygen and hydrogen with respective electronegativities of 344 and 220. New Movie Trailers Were Excited About.
Electronegativity is a periodic table trend.

What 2 elements make up a covalent compound. Your biggest friend when it comes to understanding chemical bonding is the periodic table because its organized to group elements with similar charges together for example all of the alkali metals carry a 1 charge. The stronger a bond the greater the. Only when two atoms of the same element form a covalent bond are the shared electrons actually shared equally between the atoms.
You should know the Roman numerals for 1 I 2 II 3 III 4 IV 5 V and 6 VI. The periodic table also known as the periodic table of the chemical elements is a tabular display of the chemical elementsIt is widely used in chemistry physics and other sciences and is generally seen as an icon of chemistryIt is a graphic formulation of the periodic law which states that the properties of the chemical elements exhibit a periodic dependence on their atomic numbers. While there are higher oxidation numbers they are less common.
Mineral - mineral - Classification of minerals. Under this scheme they are divided into classes according to their dominant anion or anionic group eg halides oxides and sulfides. In many cases two elements can combine in several different ways to make completely different compounds.
Chemical compound any substance composed of identical molecules consisting of atoms of two or more chemical elements. Understand a covalent compound. Since the middle of the 19th century minerals have been classified on the basis of their chemical composition.
All the matter in the universe is composed of the atoms of more than 100 different chemical elements which are found both in pure form and combined in chemical compoundsA sample of any given pure element is composed only of the atoms characteristic of that element and. The name of an ionic compound also gives you information about the oxidation numbers of the elements. The name for the compound is based off how many atoms are present.
We measure the strength of a covalent bond by the energy required to break it that is the energy necessary to separate the bonded atoms. These effects add as vectors to make the overall molecule polar. Water consists of compound molecules made up of 2 hydrogen atoms and 1 oxygen atom.
About one-quarter of the elements are also found in the free state. A majority of elements on earth are found in chemical combinations with other elements. Two nonmetals combine to form a covalent or molecular compound ie one that is held together by covalent bonds which result from the sharing of electrons.
The electronegativity difference polarizes each HO bond shifting its electrons towards the oxygen illustrated by red arrows. This is why its called H 2 O. Stable molecules exist because covalent bonds hold the atoms together.
When atoms of different elements share electrons through covalent bonding the electron will be drawn more toward the atom with the higher e lectronegativity resulting in a polar covalent bond. Eleven elements make up about 99 of the earths crust and atmosphere. Separating any pair of bonded atoms requires energy see Figure 1 in Chapter 72 Covalent Bonding.
Oxygen constitutes nearly one-half and silicon about one-quarter of the total quantity of these elements. Several reasons justify use of this criterion as the distinguishing factor at the highest level. Atoms with the same electronegativity form nonpolar covalent bonds.
The prefix attached is the Latin term for the number of molecules. Water will always have 2 times the number of hydrogen atoms as oxygen atoms. Covalent compounds are formed by two or more nonmetal elements.
When compared to ionic compounds covalent. When atoms of different types of elements join together they make molecules called compounds. Take A Sneak Peak At The Movies Coming Out This Week 812 New Movie Releases This Weekend.

Covalent Bond Definition Properties Examples Facts Britannica

Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry
Single And Multiple Covalent Bonds Article Khan Academy
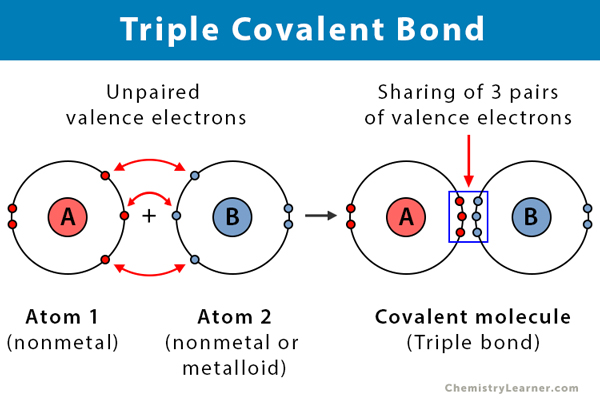
Triple Covalent Bond Definition And Examples
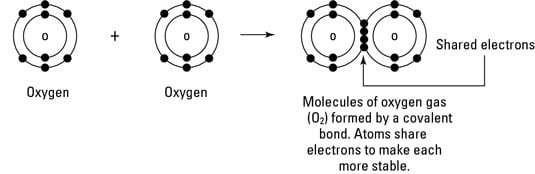
Environmental Science What Is Covalent Bonding Dummies

How To Predict Number Of Bonds Each Element Forms Chemsimplified
Covalent Bond Ck 12 Foundation

Molecules And Compounds Ppt Video Online Download
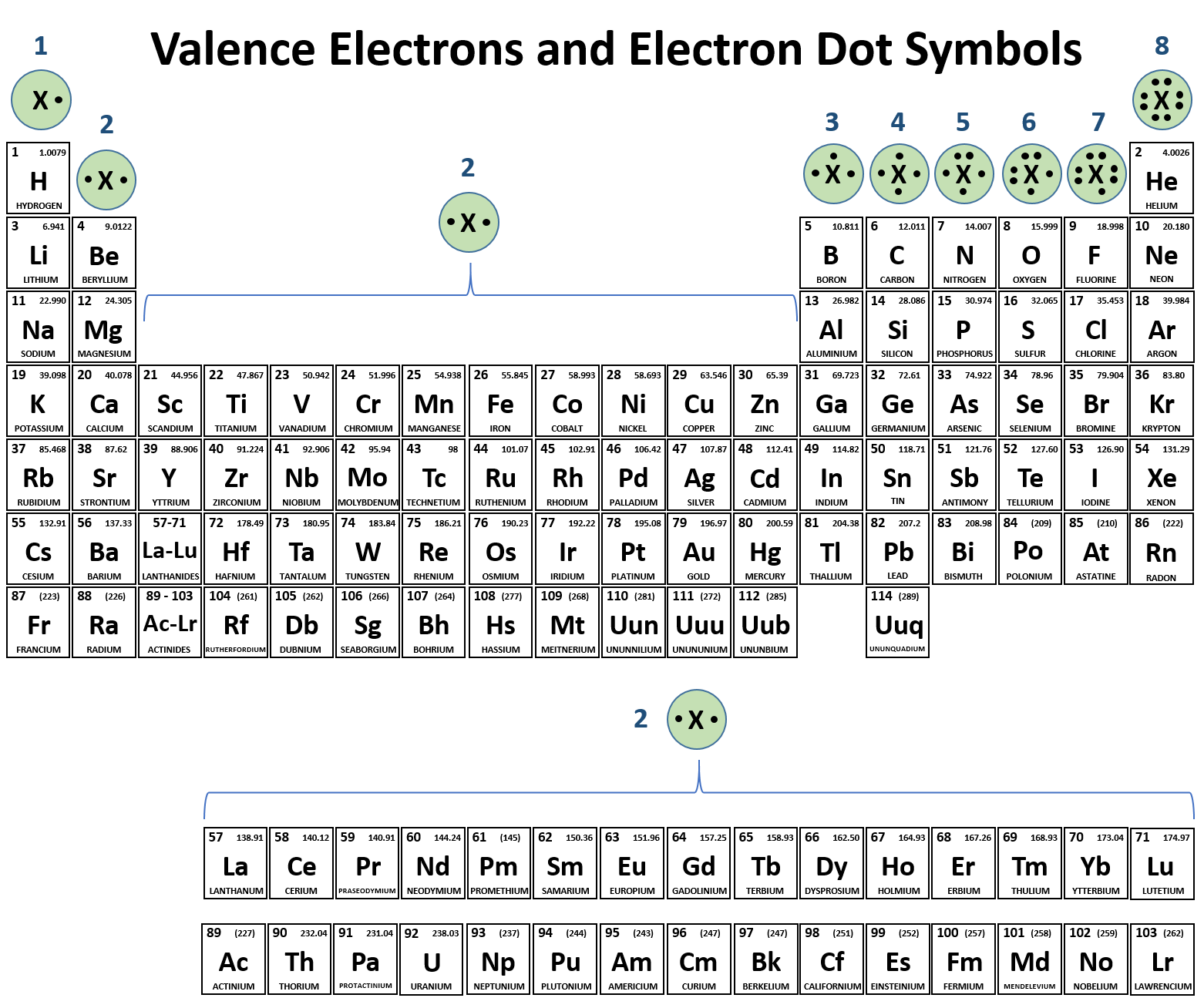
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry
Single And Multiple Covalent Bonds Article Khan Academy

Naming Covalent Compounds Nomenclature Rules
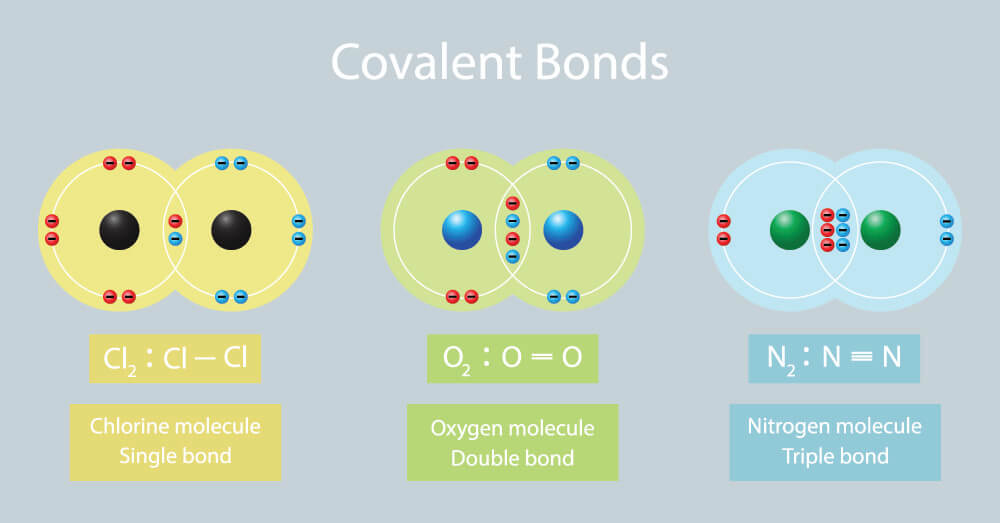
Covalent Bond Biology Dictionary

Covalent Bonds Biology For Majors I

Covalent Bond An Overview Sciencedirect Topics

Covalent Compounds Manoa Hawaii Edu Exploringourfluidearth
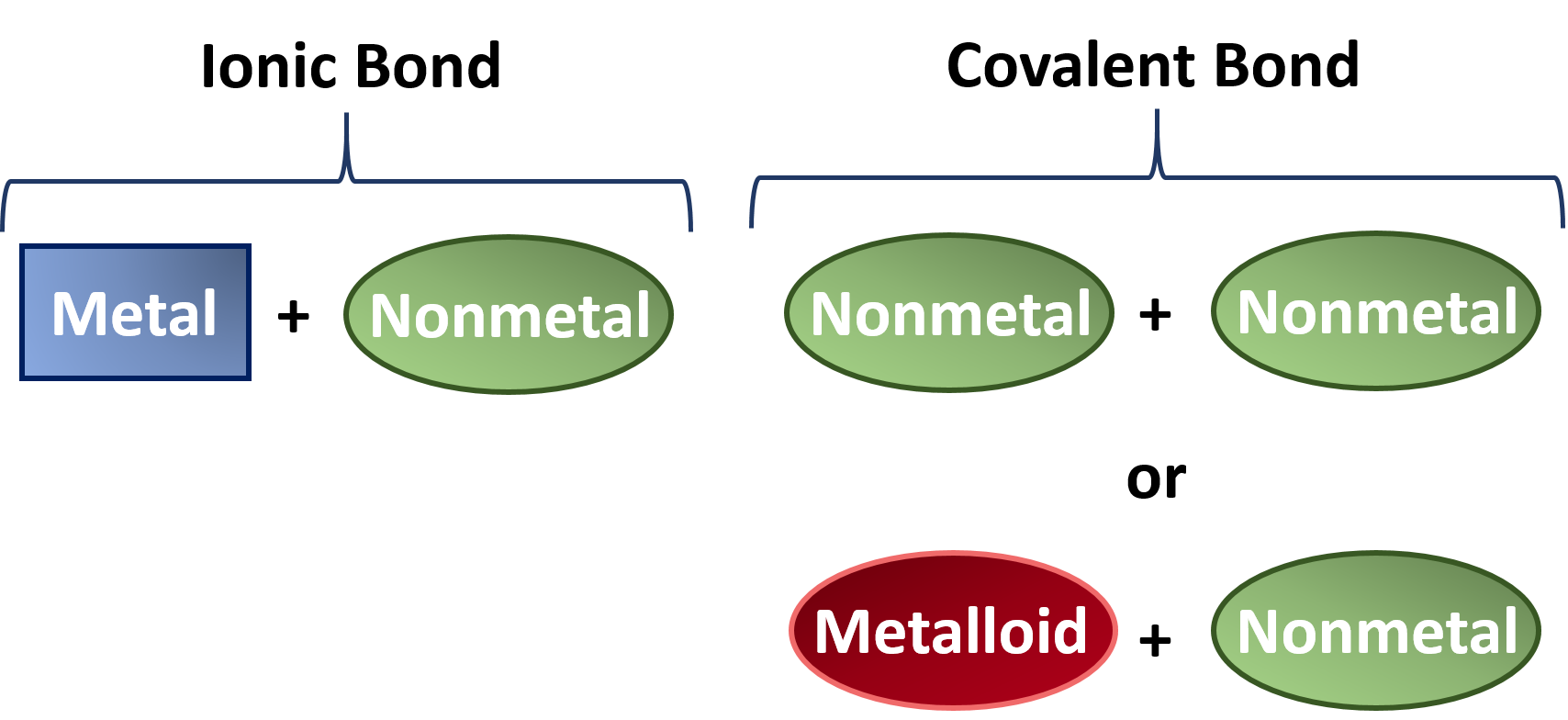
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry
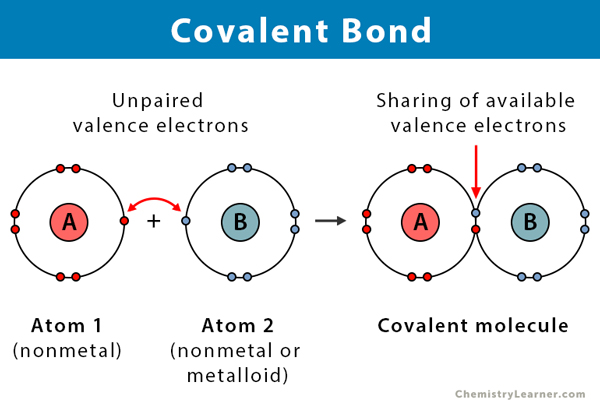
Covalent Bond Definition Types And Examples
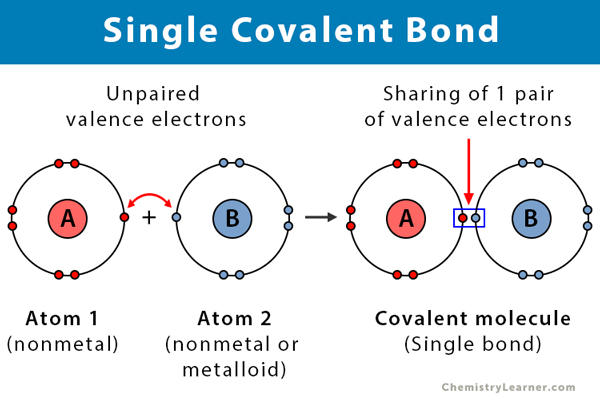
Single Covalent Bond Definition And Examples
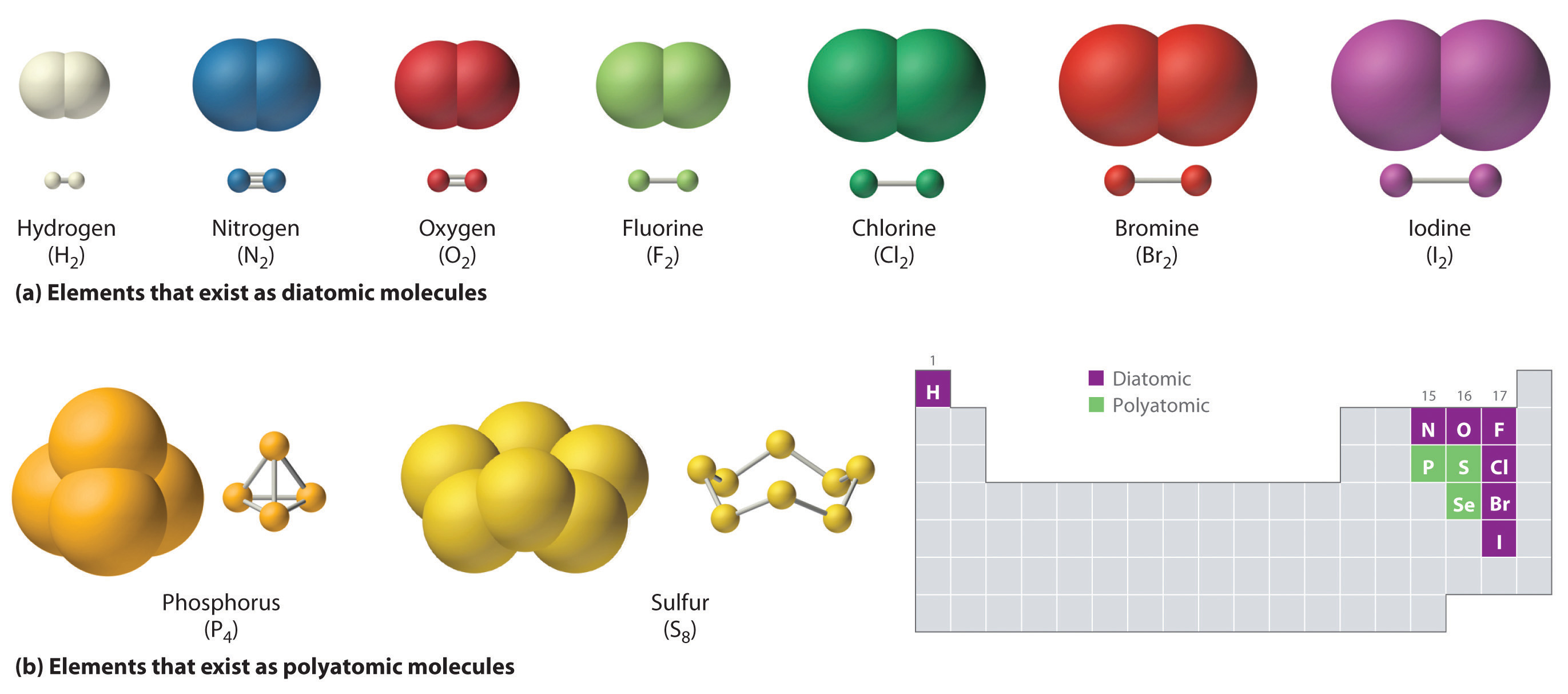
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry


